Carbonyl Compounds
Welcome to Carbonyl Crescent – a small, overachieving street where every resident has a carbonyl group (C=O) in their backbone and far more chemical personality than a plain hydrocarbon. Add C=O to a carbon chain, and you change its polarity, reactivity and how it behaves in plant chemistry, from sharp fruit acids to fragrant esters and sturdy amide links in proteins.
On this page, we’re walking down Carbonyl Crescent to meet the main families you’ll keep seeing in both organic chemistry and herbal medicine: aldehydes (–CHO), ketones (>C=O), carboxylic acids (–COOH), esters (–COOR) and amides (–CONH–). Each retains the same C=O core, but what’s attached to it changes everything, including how it dissolves, how it reacts, and where it appears in herbs, oils, and essential oils.
Aldehyde
Aldehydes: No. 1 Carbonyl Crescent (–CHO)

No. 1 Alde-House, Carbonyl Crescent: The scruffy end-of-terrace aldehyde. The grumpy molecule in the window has a carbonyl (C=O, red and grey) with a single hydrogen (H) and an R group on the same carbon – that makes it an aldehyde (–CHO). It’s literally at the end of the street because aldehydes have their carbonyl at the end of the chain and are more exposed and reactive than ketones. The peeling paint is a reminder that an aldehyde is reactive and only one oxidation step away from saying “f**k it” and going to COOH & Chips down the road and turning into a carboxylic acid (–COOH) for a chippy tea.
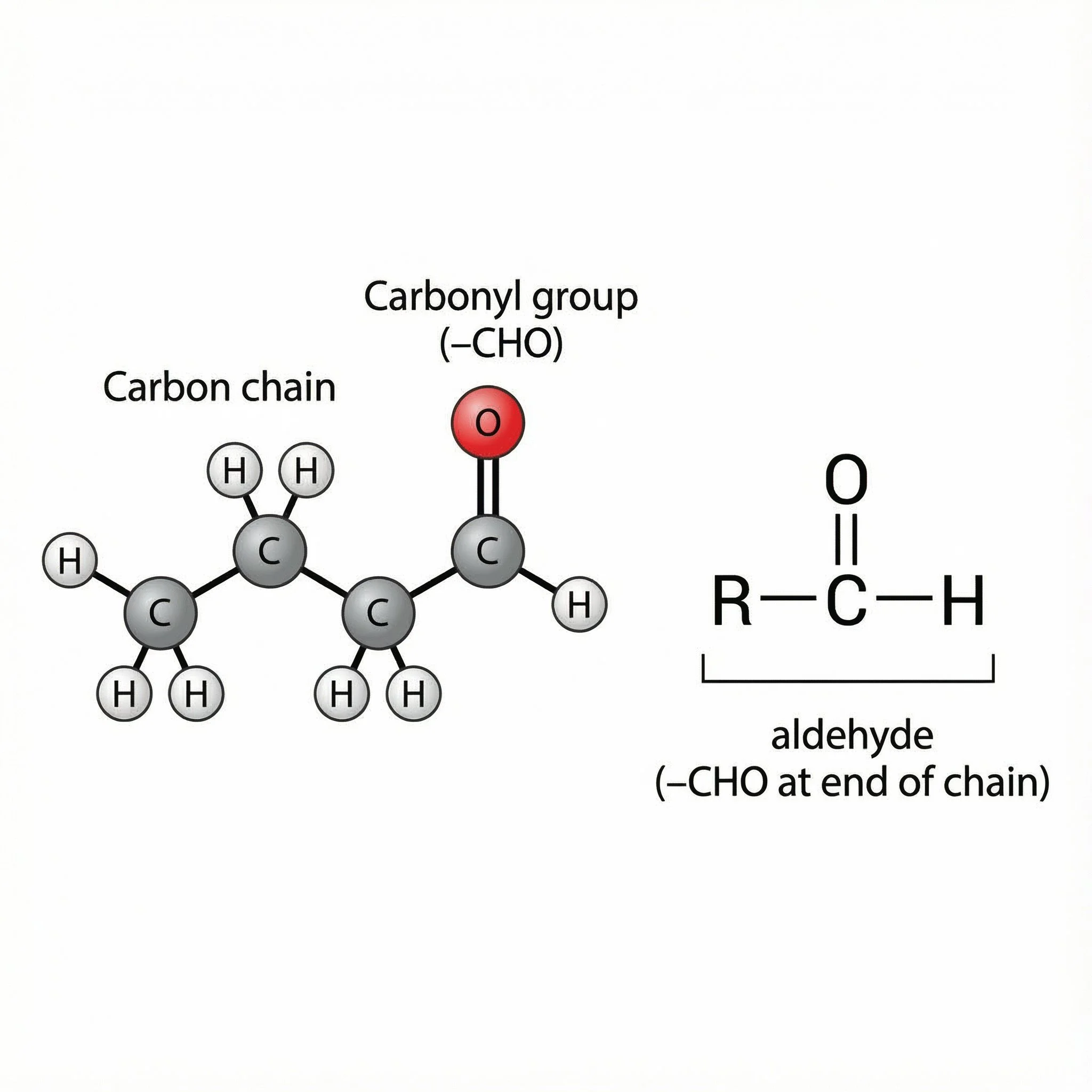
An aldehyde is a carbonyl compound where the C=O group is at the end of a carbon chain, and the carbonyl carbon is bonded to:
one hydrogen, and
one carbon group (R)
General pattern:
R–C(=O)H
often written as –CHO
On Carbonyl Crescent, aldehydes live in the end-of-terrace house at No. 1. That “end-of-terrace” idea is your reminder that:
the carbonyl is at the end of the chain, and
It’s a bit more exposed and reactive than when it’s in the middle (like in a ketone).
The “slightly scruffy” part is the memory hook: an aldehyde is often one oxidation step away from becoming the smarter, more “finished” carboxylic acid on the corner at COOH & Chips.
How to spot an aldehyde
Look for:
A C=O at the end of the molecule
The carbonyl carbon is attached to H on one side and R on the other
A formula or shorthand ending in –CHO
Naming pattern:
Names usually end in “-al”
e.g. methanal, ethanal, propanal, butanal
What aldehydes do
The main points:
More reactive than ketones
Less crowded around the carbonyl (one side is just H), so reagents can get in more easily.
Easily oxidised to carboxylic acids
Aldehyde → carboxylic acid (e.g. ethanal → ethanoic acid).
This is why classic tests (Tollens’, Fehling’s/ Benedict’s) pick up aldehydes but not ketones.
Can be reduced to primary alcohols
Aldehyde ⇌ primary alcohol (depending on reagents/direction).
Often strong-smelling
Short ones can be pungent; many aromatic/longer aldehydes are key flavour and fragrance molecules.
Aldehydes in plant / herbal chemistry
You’ll meet aldehydes in:
Essential oils and aromas – e.g.
Cinnamaldehyde (cinnamon)
Citral (lemony smell in some citrus and lemongrass)
Vanillin (main aroma of vanilla – an aromatic aldehyde)
They’re usually volatile and reactive, contributing to a herb’s scent, flavour, and sometimes irritant or stimulating effects.
Ketone
Ketone Cottage - C=O in the middle.

Ketone Cottage – C=O in the middle. Our smug-faced neighbour is a ketone: the carbonyl carbon (grey C with red O on top) is bonded to two carbon groups, R and R′, and no hydrogen, so it lives safely in the middle of Carbonyl Crescent. That comfy position is why ketones are more stable and harder to oxidise than the scruffy aldehyde at No. 1, who’s only one step away from moving into COOH & Chips and becoming a carboxylic acid. For revision:
Middle of chain, R–C(=O)–R′ = ketone
Formed from secondary alcohols
Less keen to oxidise than aldehydes, often turning up as aroma molecules in plants and essential oils rather than sour acids.
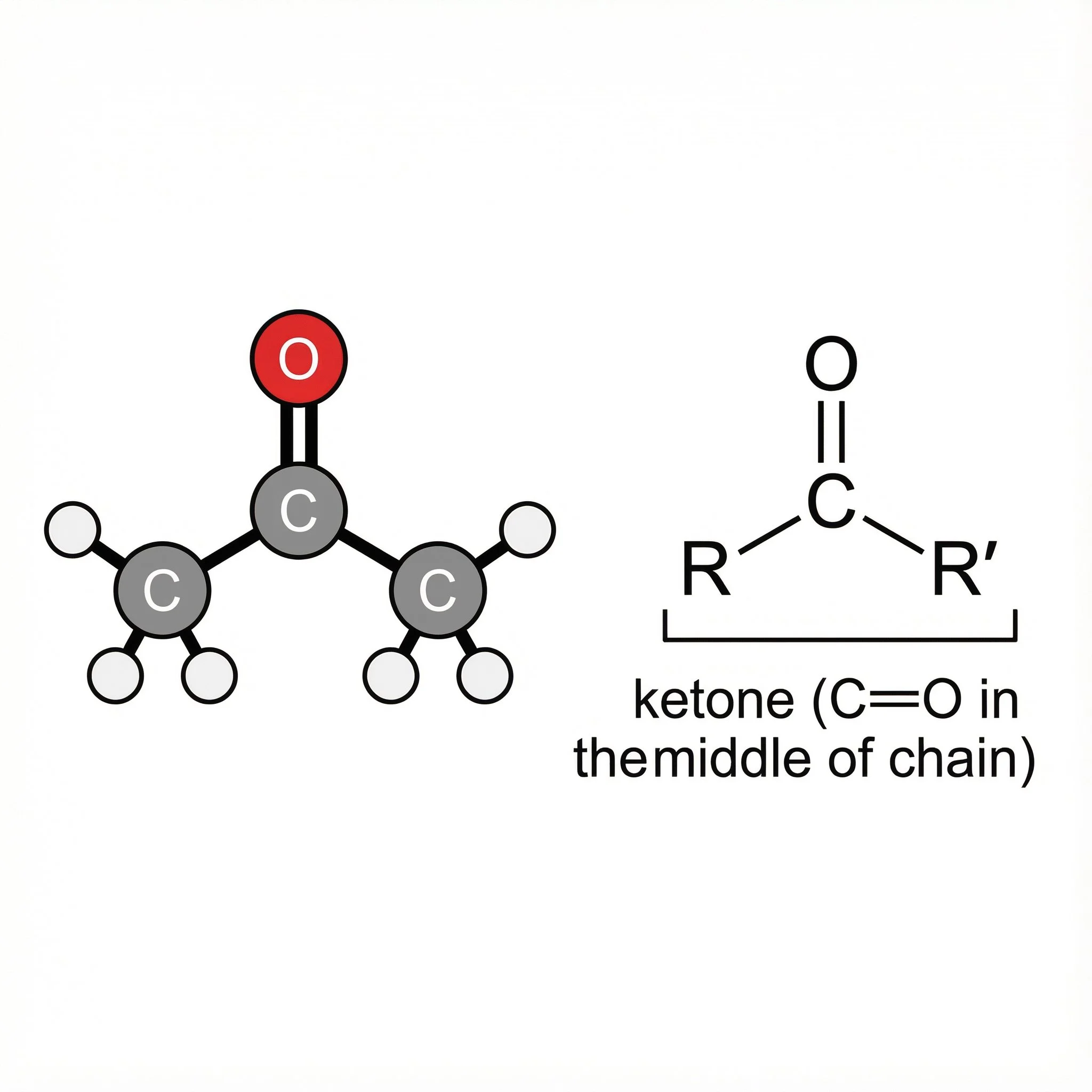
A ketone is a carbonyl compound where the C=O group sits in the middle of a carbon chain. The carbonyl carbon is bonded to:
two carbon groups (R and R′)
no hydrogens on the carbonyl carbon
General pattern:
R–C(=O)–R′ (often written as >C=O)
On Carbonyl Crescent, ketones live in the middle terrace – neighbours (carbons) on both sides, no exposed hydrogen at the end. That’s your visual cue that ketones are:
more “shielded” around the C=O
harder to oxidise than the scruffy aldehyde at No. 1 (–CHO).
Revision bullets points
Spot: C=O in the middle of the chain, attached to two carbons, no –H on that carbon.
Naming: usually ends in “-one” – propanone, butanone, cyclohexanone.
Made from: oxidation of a secondary alcohol.
Reactions: can be reduced back to a secondary alcohol; undergo nucleophilic addition at C=O; don’t oxidise further easily (unlike aldehydes).
Ketones in Herbal Medicine
In plant chemistry, ketones are a diverse bunch. They all share the same core – a carbonyl group in the middle of a carbon chain (R–C(=O)–R′), but their effects range from gentle and aromatic to quite punchy and stimulating.
You’ll most often meet ketones in:
Essential oils and volatile fractions
– e.g. camphor (from Cinnamomum camphora), carvone (caraway, spearmint), menthone (mints), and thujone (sage, some Artemisias).
– These contribute to the characteristic scent of many herbs, minty, camphoraceous, “nose-opening” aromas.Flavour compounds
– Some ketones help create spicy, herbal and minty flavours, shaping a herb’s overall sensory profile and how people experience the remedy.
From a herbalist’s point of view, ketones tend to be:
Volatile and lipophilic: more at home in essential oils, inhalations and aromatic rubs than in simple watery infusions.
Often stimulating or clearing in feel: think “opens the head and sinuses”, “sharp, penetrating smell”.
Sometimes dose-sensitive: a few ketone-rich essential oils are linked with neurotoxic or convulsant risks in high doses, so they’re used carefully and in small amounts.
And do they react? Yes, but in a calmer, more refined manner than aldehydes. The carbonyl in a ketone is still polar, so that the carbonyl carbon can take part in nucleophilic addition and reduction reactions, and it can be reduced back to a secondary alcohol. But with carbon neighbours on both sides and no hydrogen on the carbonyl carbon, ketones are less exposed and more challenging to oxidise than the scruffy aldehyde at No. 1 Carbonyl Crescent. They still “do chemistry”; they don’t rush off to COOH & Chips at the first sign of an oxidising agent.
Carboxylic acids
COOH and Chips.

COOH & Chips: the carboxylic acid end of Carbonyl Crescent.
Behind the counter is our carboxylic acid molecule: a central grey carbon with a red C=O on top and a red O–H to the side, both on the same carbon, plus a blue R tail – the classic –COOH functional group. The chalkboard explains: “–COOH = C=O + O–H on the same carbon”, with everyday examples on the menu, vinegar (ethanoic acid) on your chips, lemon (citric acid) and frying oil (long-chain fatty acids).
Through the window, you can see the rest of Carbonyl Crescent: Aldehyde House at one end (–CHO, easily oxidised) and Ketone Cottage in the middle (R–C(=O)–R′, harder to oxidise). COOH & Chips marks the fully oxidised stop on the street, and is your reminder that whenever you see –COOH, you’re looking at a carboxylic acid related to the acids in fruit, vinegar and fatty oils.
Carboxylic Acids - COOH and CHIPS.
I have used the “COOH and CHIPS” shop as a study aid of carboxylic acids: ethanoic acid in vinegar on chips, citric acid in the lemon squeezed over fish, and long-chain fatty acids in the frying oil. Your COOH & Chips mascot is a reminder that all of these share the same –COOH “heart”, even though their chains and behaviour differ.
Carboxylic acids are molecules with the –COOH functional group – a carbonyl (C=O) and a hydroxyl (O–H) attached to the same carbon, written R–C(=O)–OH. This arrangement makes them weak organic acids that are usually relatively polar and often sour-tasting.
In herbal medicine, the same family shows up as fruit acids (citric, malic, tartaric), lactic acid in fermented preparations, and fatty acids in herbal oils and balms. They help shape a remedy’s taste (e.g., sharp, tangy), water solubility, tissue interactions, and skin-barrier effects, making the –COOH group an important part of a herb’s overall chemical character.
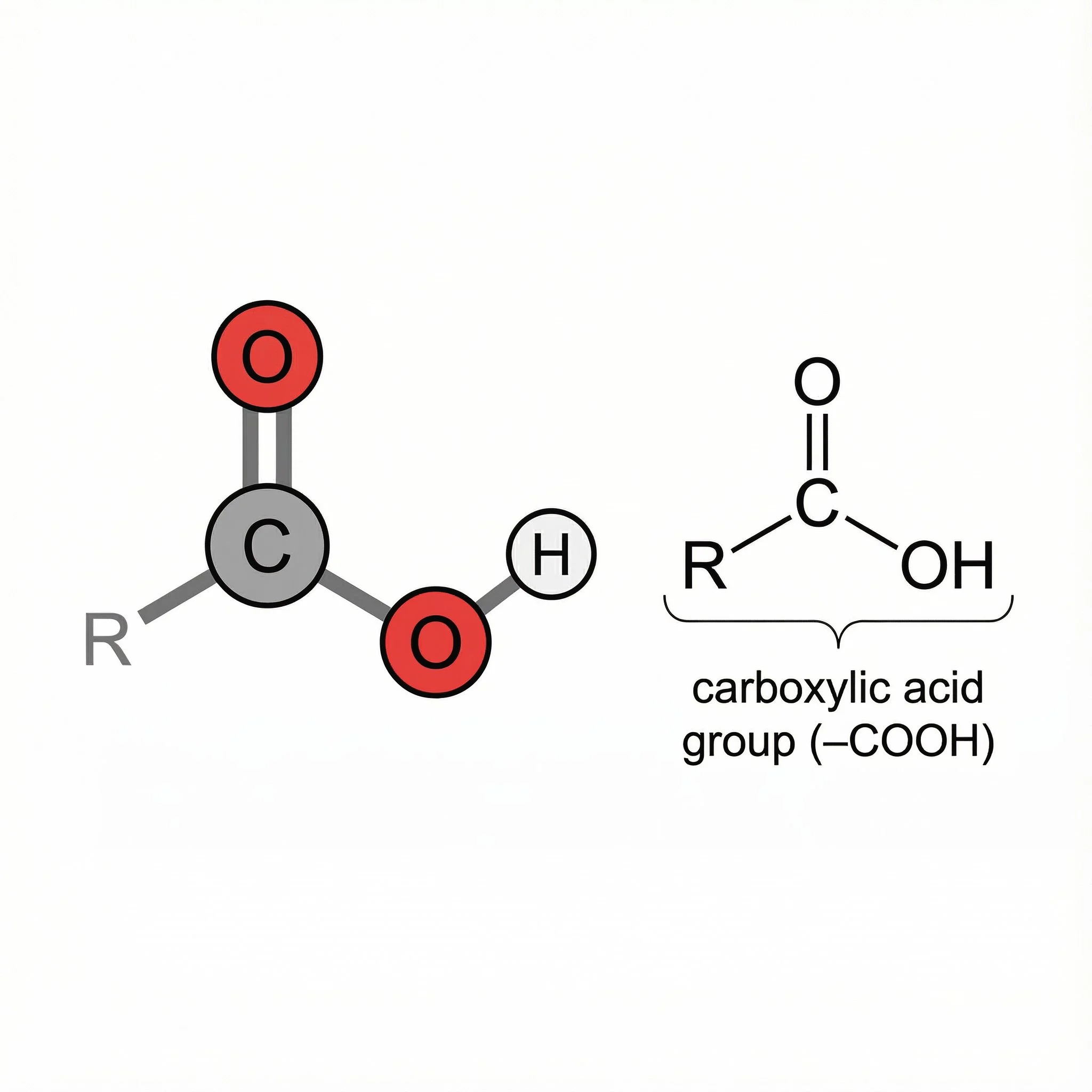
A closer look at –COOH.
The –COOH group has two highly electronegative oxygen atoms attached to a carbon atom, so they draw electron density toward themselves. This makes the O–H bond in –COOH easier to break, so the group can lose a proton (H⁺) and behave as an acid. When the proton is lost, you get –COO⁻, and the negative charge is shared between the two oxygens – this helps to stabilise the ion and makes carboxylic acids more acidic than alcohols. The –COOH group is also polar and can form hydrogen bonds, which gives small carboxylic acids relatively high boiling points and good solubility in water. In herbal chemistry, polarity and acidity help explain why fruit and phenolic acids readily extract into teas and tinctures, whereas fatty acids behave differently in oils and on the skin.
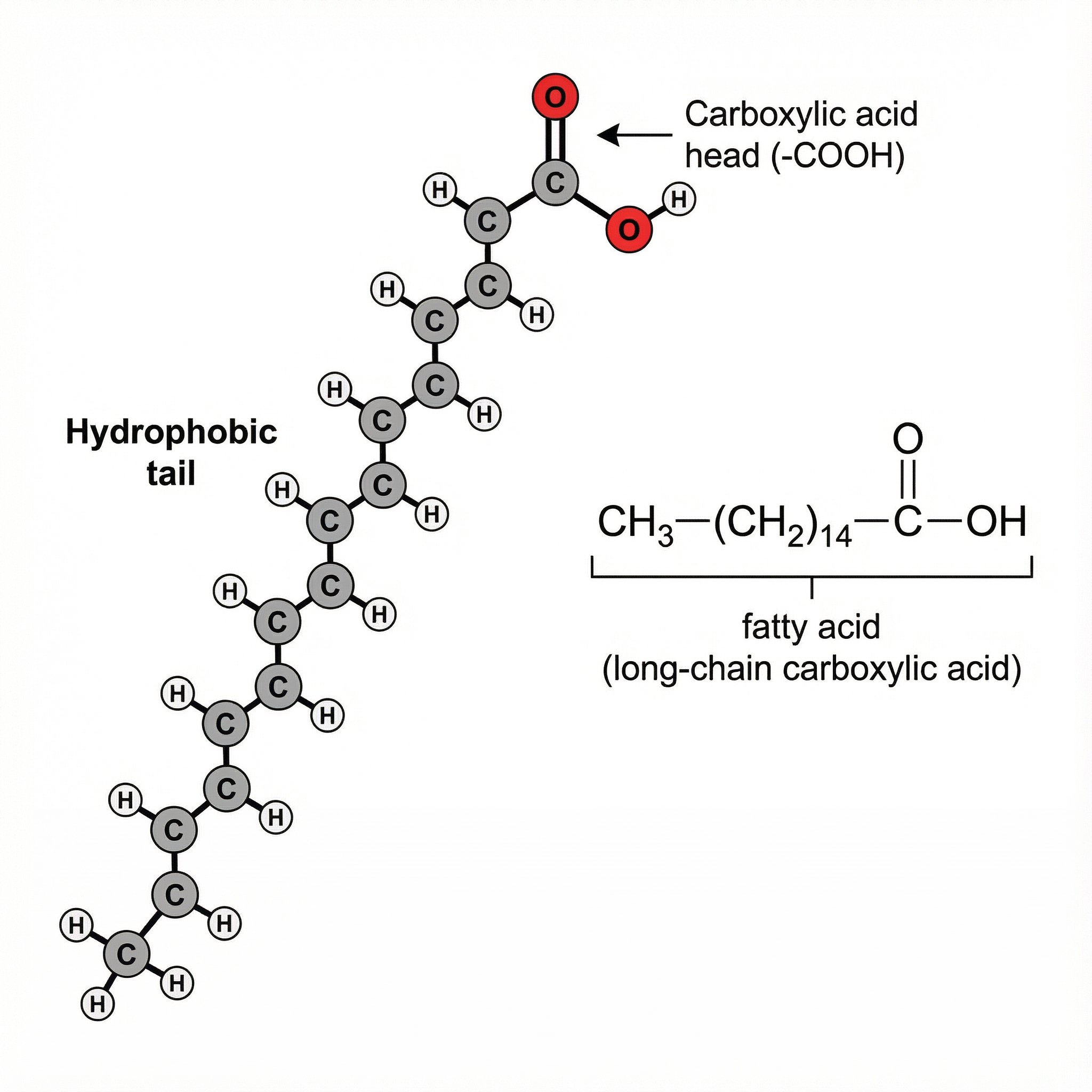
Fatty Acids – Long-Tailed Carboxylic Acids
Fatty acids are basically carboxylic acids with a long hydrocarbon tail. Chemically, they still have the same –COOH group at one end, but instead of a tiny R group, they’ve got a long chain of carbons and hydrogens:
R–C(=O)–OH, where R is a long carbon chain (often C₁₂–C₂₂ or more).
At one end, you’ve got the polar, acidic –COOH “head”, and at the other a non-polar, oily “tail”. That mix renders fatty acids amphipathic; one end prefers water, whereas the rest interacts with oils and membranes.
You’ll see them described as:
Saturated: no C=C double bonds, straight tails, pack tightly (e.g. stearic acid).
Unsaturated: one or more C=C double bonds, kinked tails, more fluid (e.g. oleic, linoleic).
Fatty acids, COOH & the chip shop link
In the COOH & Chips world, fatty acids are the ones hiding in the frying oil: still carboxylic acids, just with very long tails. The –COOH head is the same as the fella in the chip shop, but most of the molecule is a greasy hydrocarbon chain.
That’s why:
They don’t dissolve in water (the tail dominates),
But they mix beautifully with oils, membranes and skin,
And form the basis of triglycerides (three fatty acids + glycerol).
Why fatty acids matter in herbal medicine
From a herbalist’s point of view, fatty acids are essential because they:
Make up the fixed oils in seeds and nuts (e.g. evening primrose, flax, sunflower).
Contribute to skin-barrier repair and emollient effects in creams, balms and infused oils.
Affect inflammation and cell signalling (especially essential fatty acids like omega-3 and omega-6).
Explain why some constituents only really show up in oil-based preparations (infused oils, ointments) rather than teas and tinctures.
So:
Same –COOH family,
But with a long hydrophobic tail that pulls them into the world of oils, membranes and skin medicine, rather than your teapot.
Ester
Bakery & Florist.

Ester’s Bakery & Florist – fruity and floral carbonyls.
At the pretty end of Carbonyl Crescent, Ester runs a shop that sums up what esters do: her molecule body is R–C(=O)–O–R′, a carbonyl (C=O) joined to an –O–R′ group, made when a carboxylic acid and an alcohol combine. The bakery side, apple turnovers (apple esters like ethyl butanoate) and banana pastries (isoamyl acetate), nod to the fruity smells of many simple esters. On the florist side, lavender and bergamot, loaded with linalyl acetate, plus other bouquets labelled “floral esters”, show that esters also impart soft, perfume-like aromas in herbs and essential oils.
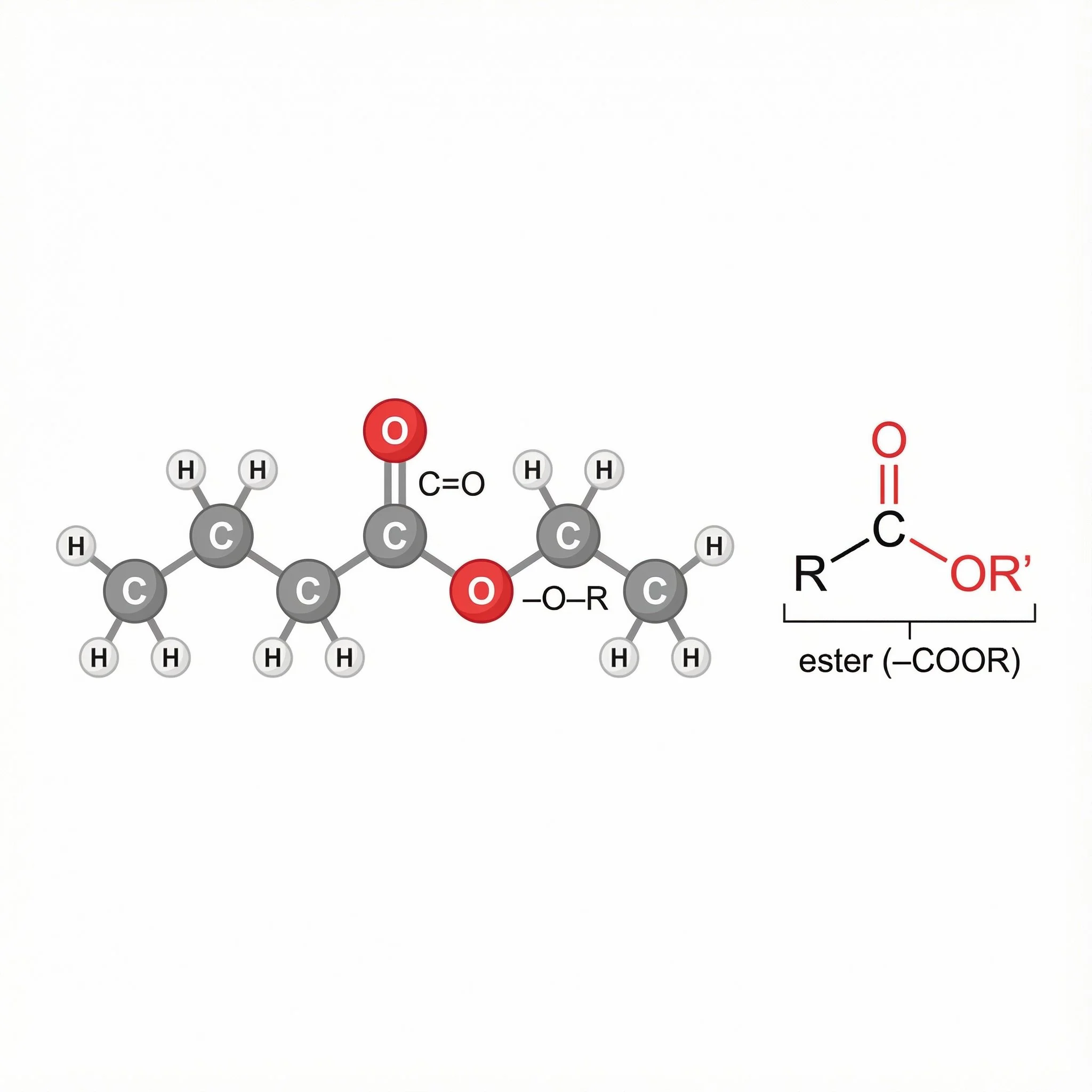
Esters: Acid + Alcohol in Disguise
On Carbonyl Crescent, esters are the ones who’ve tidied themselves up and opened a pretty shop. Chemically, an ester is what you get when a carboxylic acid and an alcohol react together and lose water:
carboxylic acid + alcohol ⇌ ester + water
The functional group is:
R–C(=O)–O–R′
The R–C(=O) bit comes from the acid.
The –O–R′ bit comes from the alcohol.
At A-level, key points are:
Structure: carbonyl (C=O) next to an –O–R′, so it’s not –COOH anymore, it’s –COOR′.
Naming:
the alcohol part becomes the “-yl” (e.g. ethyl)
the acid part becomes the “-oate” (e.g. ethanoate)
→ ethyl ethanoate, isoamyl acetate, etc.
Formation (esterification): heat a carboxylic acid with an alcohol and a strong acid catalyst (often conc. H₂SO₄) to get an ester + water.
Hydrolysis: you can go back the other way; under acidic or alkaline conditions, esters can be split back into the acid + alcohol.
Why do esters smell so nice?
Because they’re:
reasonably volatile (they evaporate into the air easily)
often less polar than the parent acids, so they don’t cling to water as much
small enough (in many cases) to reach your nose quickly
Short-chain esters tend to smell fruity (apple, pear, banana, pineapple), while many terpene esters in essential oils smell floral or perfumey (lavender, bergamot, rose-like notes).
So Ester’s shop is your memory hook:
Bakery side = fruity esters (apple, banana, “yummy” smells).
Florist side = floral esters (lavender, bergamot, bouquets and perfumes).
And behind it all, the same chemistry: acid + alcohol → R–C(=O)–O–R′ + H₂O..
Esters in Herbal Medicine
In plant chemistry, esters are often the nice-smelling side of the family. Because they’re volatile and fairly lipophilic, they turn up a lot in essential oils and can make a herb smell soft, fruity, floral or “perfumey”.
Common examples you’ll meet in herbal texts include:
Linalyl acetate – a major ester in lavender, bergamot and clary sage oils; often linked with their gentle, relaxing, “soft” aroma.
Bornyl acetate – found in conifers (pine, fir, spruce) and some aromatic herbs, giving fresh, resinous notes.
Geranyl and citronellyl acetates – in rose, geranium and related oils, contributing to rosy-floral scents.
Various fruity esters in fennel, anise, chamomile and citrus peels, adding sweetness and roundness to the aroma.
From a herbalist’s point of view, esters tend to be:
Aromatic and volatile – more relevant to inhalations, diffused oils, balms and massage blends than to simple teas.
Often described in herbal literature as having gentler, smoothing or antispasmodic qualities compared with sharper, more “aggressive” constituents (though the exact effects depend on the whole plant and preparation, not just one ester).
A clue that a herb’s smell and “feel” may be part of its traditional use – for example, calming lavender pillows, soothing chamomile, or uplifting citrus oils.
So in Carbonyl Crescent terms:
COOH & Chips handles the sour, properly acidic end.
Ester’s Bakery & Florist is where those acids have teamed up with alcohols, toned things down, and turned into the fruity jams and floral bouquets of the herbal world.
Amide
Amide House – Neighbourhood Watch.

Amide House – CONH in the middle, keeping an eye on everyone.
This slightly smug neighbour is an amide: a central grey carbon with a red C=O on top, a blue N with two white H atoms on one side (–CONH₂) and a blue R tail on the other, the classic R–C(=O)–NH₂ group. He’s swapped the carboxylic acid’s –OH for –NH₂, put on a cardigan and hi-vis, and joined the Neighbourhood Watch, Amides.
On Carbonyl Crescent, aldehydes up the road are more reactive and easily oxidised, ketones are calm in the middle, COOH & Chips and Ester florist and Bakery are fully acidic at the corner, and Amide House is the stable one, harder to hydrolyse, good at hydrogen bonding, and in biochemistry the sort of linkage that holds proteins and peptide chains together.
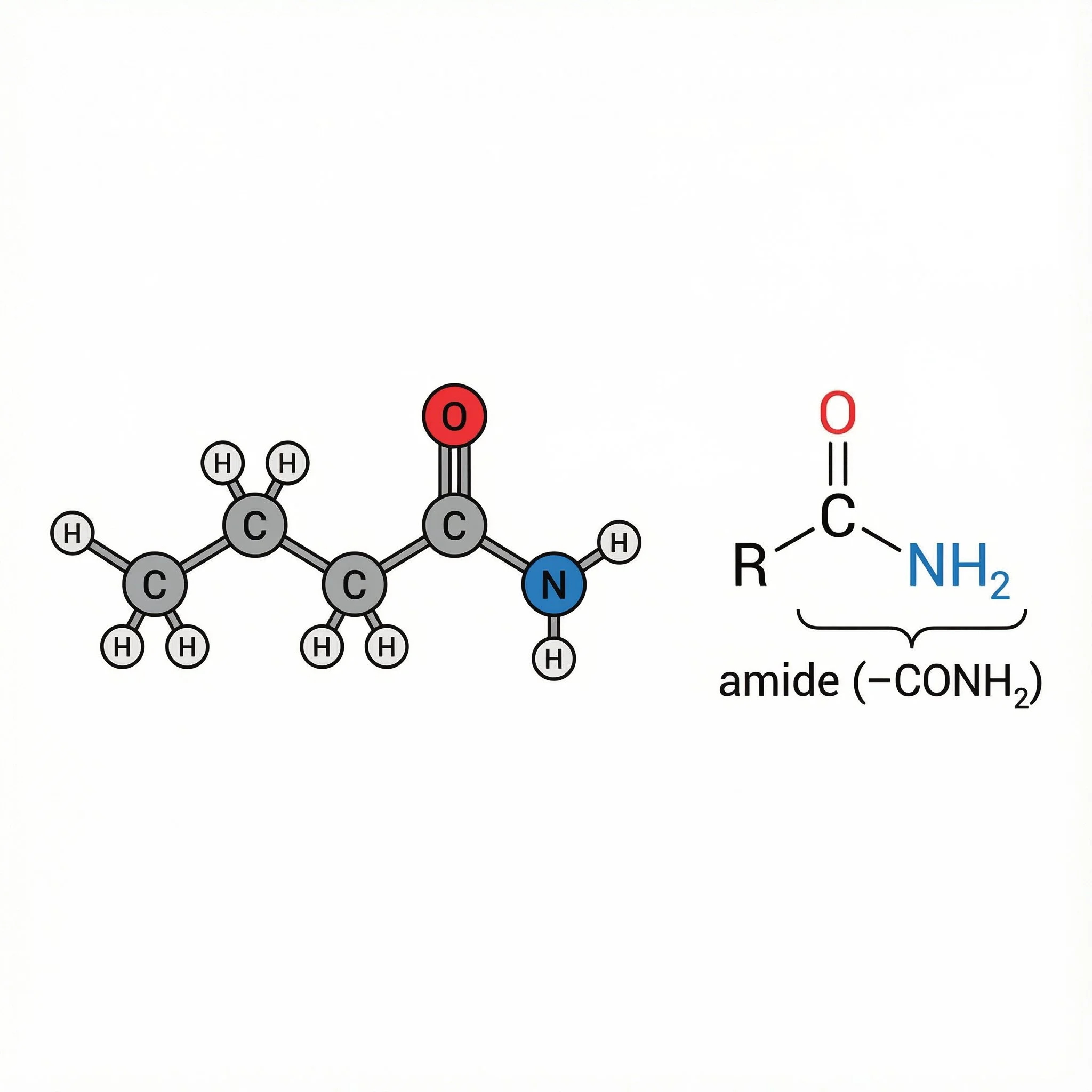
What is an amide?
Chemistry first:
General structure: R–C(=O)–NH₂, R–C(=O)–NHR or R–C(=O)–NR₂
In other words, take a carboxylic acid and swap the –OH for –NH₂ / –NHR / –NR₂:
carboxylic acid - replace –OH with –NH₂ - amide (–CONH–)
On Carbonyl Crescent that’s the difference between COOH & Chips (acid) and Amide House (the same carbonyl, but now linked to nitrogen).
Key features
The carbonyl carbon is still C=O, but the lone pair on nitrogen can delocalise towards the carbonyl.
The C–N bond is partly double in character.This resonance makes amides:
More stable and less reactive than acids, esters or acyl chlorides
Poor bases (much less basic than amines)
Able to hydrogen bond strongly (N–H and C=O both take part)
Consequences:
Higher boiling points than similar amines or esters (lots of H-bonding).
Small amides (e.g. acetamide) can be quite water-soluble; bigger ones become more organic-solvent-loving.
They do hydrolyse back to acids + amines, but usually only with strong acid or base and heat – hence our slightly smug neighbour: he’s not easily pushed around.
How to recognise an amide
Look for:
A carbonyl (C=O)
Immediately next door to a nitrogen (N with H’s or carbon groups attached)
–CONH– or –CONH₂
Amides in herbal / biological chemistry
Even though you won’t see “amide” shouted on every herb label, they’re everywhere in living systems, so they’re quietly important for herbal medicine.
Peptide bonds and plant proteins
Every time two amino acids link to form a dipeptide, polypeptide or protein, the bond between them is an amide (a peptide bond, –CONH–).
Plant enzymes, storage proteins, and many receptors are full of amide bonds.
When we talk about protein-rich seeds or herbs, or enzymes affecting how constituents are formed or broken down, we’re indirectly talking about amide chemistry.
Amide-containing plant constituents
Some strongly active plant molecules that turn up in herbal or nutritional contexts are amides:
Capsaicin in Capsicum annuum (chilli) – an amide between a vanillylamine and a fatty acid–like chain. It interacts with TRPV1 pain/heat receptors, giving heat, pain, then desensitisation.
Piperine in black pepper (Piper nigrum) – another amide that affects pungency and can influence absorption of other compounds.
Various more complex alkaloids and secondary metabolites include amide fragments in their backbone, helping set their 3D shape and how they bind to receptors.
Extraction and formulation angles
Because amides:
can hydrogen bond,
but are often built into larger, more lipophilic molecules,
you’ll often find that:
Simple, small amides (or peptide fragments) are more at home in water / aqueous extracts.
Bigger, aromatic amide constituents (like capsaicin, piperine) extract better into alcohol, glycerol or oils, and show up in tinctures, infused oils, balms, liniments rather than plain teas.
That fits nicely with practice:
You don’t usually take chilli as a gentle tea, you meet capsaicin in food, rubs and liniments.
Black pepper’s piperine effect appears in spice mixes, infused oils and some tincture formulas rather than delicate infusions.
Amides = COOH that’s settled down with nitrogen, less dramatic, harder to push around, but essential for proteins, pungent amide constituents like capsaicin and piperine, and the structural chemistry behind a lot of plant and human biology.
Sources
Berg, J. M., Tymoczko, J. L., Gatto, G. J., Jr., & Stryer, L. (2019). *Biochemistry* (9th ed.). W. H. Freeman.
Bone, K., & Mills, S. (2013). *Principles and practice of phytotherapy: Modern herbal medicine* (2nd ed.). Churchill Livingstone.
CGP Books. (2021). *A-Level chemistry: Complete revision & practice (with online edition).* Coordination Group Publications.
McIntyre, A. (2019). *The complete herbal tutor: The definitive guide to the principles and practices of herbal medicine* (Rev. & expanded ed.). Aeon Books.
McMurry, J. E. (2016). *Organic chemistry* (9th ed.). Cengage Learning.

